According to the Top 10 Greatest Inventions that Completely Changed the World, the world has seen numerous inventions that have transformed how we live and interact with the environment. From the wheel to the internet, these inventions have played a crucial role in shaping our modern society. One invention that stands out among all the rest is the Periodic Table. It has been widely recognized as a cornerstone of modern chemistry and has been instrumental in advancing our understanding of the natural world. In recognition of its significance, National Periodic Table Day is celebrated annually on February 7th.
To commemorate this day, the World’s #1 Source of Research-Based Content, The Daily Top 10 thought it would be appropriate to share an article exploring the Top 10 Reasons Why We Celebrate National Periodic Table Day.

National Periodic Table Day is celebrated on February 7th every year to honor the Periodic Table’s publication by Russian chemist Dmitri Mendeleev in 1869. This invention has had a profound impact on the scientific world and remains one of the greatest achievements in the history of human discovery. The Periodic Table displays all the known elements in an organized and systematic manner, which has paved the way for numerous advancements in fields such as chemistry, physics, and biology.
So, here are the Top 10 Reasons Why We Celebrate National Periodic Table Day. But before that, check out the Top 10 Reasons Why We Celebrate National Puzzle Day, as it is also an important holiday that we celebrate.
What is the Main Reason for Celebrating National Periodic Table Day?
The Periodic Table is a tabular arrangement of chemical elements, organized based on their atomic number, electron configurations, and chemical properties. The elements are arranged in rows and columns to display periodic trends in properties such as electron affinity, ionization energy, and electronegativity. It is a valuable tool for chemists and other scientists, as it provides a systematic way of organizing and predicting the properties of the elements.
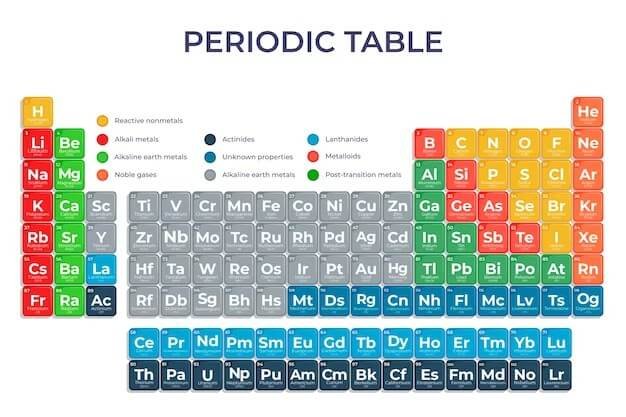
So, let’s start with all the other reasons behind celebrating National Periodic Table Day before stepping into the Main Reason Why We Celebrate National Periodic Table Day.
TOP 10: Provided a Systematic Way of Organizing Elements

The Periodic Table is a tabular arrangement of all the known elements, organized in order of increasing atomic number. Elements are grouped together based on their chemical and physical properties, such as electron configuration, reactivity, and melting and boiling points. As stated in Chemistry for Life, this systematic organization allows for predicting the properties of new elements and discovering relationships between elements.
The Periodic Table provides a convenient tool for chemists and other scientists to understand the behavior and characteristics of elements, making it a key tool in the study of chemistry. Scientists often use the Periodic Table to quickly refer to information about an element, like atomic mass and chemical symbols. Its arrangement also allows scientists to discern trends in element properties, including electronegativity, ionization energy, and atomic radius.
TOP 9: Predicted the Existence of New Elements

The Periodic Table also allowed Mendeleev to predict the existence and properties of elements that had yet to be discovered. Khan Academy mentioned that the Periodic Table arranges elements in order of increasing atomic number and grouping them by their chemical and physical properties, which repeat in a periodic manner.
This allowed scientists to predict missing elements’ properties and suggest their existence. For example, if an element had similar properties to elements in its group, scientists could predict that it would have similar chemical and physical characteristics. This led to the discovery of new elements, such as germanium, which was predicted by Dmitri Mendeleev in 1871 and later discovered in 1886.
TOP 8: Impacted Our History

According to Thought Co, the Periodic Table was born in 1868 when professor Dmitry Mendeleev needed to devise a quick way to organize the known elements for a book he was working on. On a deadline, Mendeleev decided to sort the elements by their atomic weight. An avid solitaire player, Mendeleev wrote the name and weight of each element on its own card and arranged them on a table, and the Periodic Table was born!
In 1913, Henry Moseley confirmed Mendeleev’s arrangement by establishing the relationship between an element’s atomic number and its atomic structure, providing a more accurate basis for the Periodic Table.
However, the modern Periodic Table we use today is based on Mendeleev’s original Table and Moseley’s discovery. It includes elements up to 118, arranged in rows by electron configuration and columns by chemical and physical properties.
TOP 7: Paved the Way for Modern Chemistry
The Periodic Table provided a foundation for understanding chemical reactions and the behavior of elements, leading to major advancements in the field of chemistry. As Science Direct stated, it is a visual representation of all known elements arranged according to their atomic number, electron configurations, and chemical properties. It also serves as the cornerstone of modern chemistry, providing a systematic way to understand the relationships between the elements and predict their behavior in chemical reactions.
According to Science Direct, the Periodic Table is used to classify elements into categories based on their properties, such as their reactivity, their electron configurations, and the type of bonding they form. So, this information is crucial for understanding and predicting chemical reactions, synthesizing new compounds, and developing new technologies in fields like medicine and energy.
TOP 6: Paved the Way for Understanding the Atomic Structure

The Periodic Table allowed scientists to understand the arrangement of electrons in an atom, leading to a deeper understanding of the structure of matter. The data from Western Organic University proves the Periodic Table is important because it is organized to provide a great deal of information about elements and how they relate to one another in one easy-to-use reference.
The Table can be used to predict the properties of elements, even those that have not yet been discovered. Columns (groups) and rows (periods) indicate elements that share similar characteristics. And especially makes trends in element properties apparent and easy to understand and provides important information used to balance chemical equations.
TOP 5: Influenced the Development of Quantum Mechanics

As described by the Encyclopedia, Quantum mechanics remarks the relationship between energy and matter on an atomic and subatomic scale. So, the Periodic table significantly impacted the development of quantum mechanics. The discovery of the Periodic Law by Dimitri Mendeleev provided evidence for the atomic nature of matter and paved the way for the development of modern atomic theory.
This led to the study of atomic spectra and the realization that the energies of an atom’s electrons are quantized, which was one of the key insights that led to the development of quantum mechanics. By organizing elements into the Periodic Table based on their atomic structure, Mendeleev’s work provided a foundation for understanding the behavior of atoms and molecules and their role in chemical reactions, which in turn was crucial for the development of quantum mechanics. So, the Periodic Table has played a significant role in developing quantum mechanics, a branch of physics that explores the behavior of matter and energy at a microscopic level.
TOP 4: Learned Fun Facts

National Periodic Table Day is a day to celebrate one of the most important tools in chemistry: the Periodic Table. To mark this occasion, National Day Calendar shared some fun facts about the Periodic Table that you may not have known,
- The row of elements on the far right is the “Noble Gases,” which are nearly totally inert. The elements to the far left are the “Alkaline-Earth Metals,” which are violently reactive.
- Atoms with atomic numbers higher than 92 do not exist naturally and must be man-made by bombarding different elements together.
- In 1937, Technetium became the first element to be made artificially. Nihonium, Moscovium, Tennessine, and Oganesson are the newest additions, recognized in 2016.
- Any new elements will require a brand new row to be added to the Periodic Table.
- The only two elements which occur in liquid form at room temperature are mercury and bromine.
TOP 3: Impacted the Field of Biology

According to Compound Chem, the Periodic Table had a profound impact on the field of biology, providing insight into the fundamental building blocks of life and the molecular structure of proteins and DNA. Similarly, it plays a crucial role in the field of biology by providing a framework for organizing and understanding chemical elements and their properties. The elements in the Periodic Table form the basis for all of the molecules that make up living organisms, such as proteins, DNA, carbohydrates, and lipids.
The Periodic Table provides information about the atomic structure, electronegativity, and reactivity of the elements, which helps in understanding the behavior and interactions of the molecules in biological systems. Moreover, the arrangement of elements in the Periodic Table reflects their electron configurations and allows for predictions about their chemical properties, which is useful for drug design and other biological applications. So, the Periodic Table is a valuable tool for biologists to study and understand the underlying chemical processes that drive biological systems.
TOP 2: Played a Role in the Development of New Technologies
The Periodic Table is a fundamental tool in the development of new technologies as it provides a comprehensive view of the chemical elements and their properties. According to Hitecher, it has been instrumental in developing numerous technologies, including fertilizers, plastics, and pharmaceuticals. By arranging elements based on their atomic structure, scientists can predict their chemical and physical properties, which is essential for developing new materials and technologies.
The Periodic Table also provides insight into how elements interact and form compounds, which is crucial for understanding and controlling chemical reactions. This knowledge is applied in developing new technologies, such as renewable energy, fuel cells, and new medicines. Additionally, the Periodic Table has guided the discovery of new elements which can be used in various technologies, such as nuclear power and radiography. So, it is considered a fundamental tool for scientists and engineers in developing new technologies, allowing them to make informed decisions and guide their research efforts.
TOP 1: Inspired a New Generation of Scientists

ThePeriodic Table has inspired countless scientists, students, and educators and continues to be a source of wonder and inspiration for future generations. As mentioned in Science Learn, with its organized arrangement of elements and their properties, it has been a source of inspiration for scientists for over a century. It has also served as a visual representation of the building blocks of matter and their interactions, making it easier for scientists to understand the underlying principles of chemistry.
The Periodic Table has encouraged scientists to think about the relationships between elements and how they can be used in various applications. Would you believe that the Periodic Table sparked the imagination of young scientists, inspiring them to pursue careers in the field of chemistry and other related sciences? Of course, with its iconic and recognizable design, the Periodic Table has become a symbol of scientific discovery and innovation, inspiring a new generation of scientists to explore the mysteries of the universe and push the boundaries of human knowledge. Accordingly, the main reason for Celebrating National Periodic Table Day is it Inspired a New Generation of Scientists.
What are the Main Reasons for Celebrating National Periodic Table Day?
National Periodic Table Day is a significant event that celebrates the creation and importance of the Periodic Table of elements. It is a day to recognize the contributions of Dmitri Mendeleev and other scientists who helped in its development. The ten reasons highlighted above showcase the importance of this day, including the role of the Periodic Table in our daily lives, its use in various fields, and its impact on scientific advancements. So study and learn the Periodic Table as a fun time activity and make it to the Top 10 Fun Things People Born in February Should do.
Here is the list of the Top 10 Reasons for Celebrating National Periodic Table Day:
- Inspired a New Generation of Scientists
- Played a Role in the Development of New Technologies
- Impacted the Field of Biology
- Learned Fun Facts
- Influenced the Development of Quantum Mechanics
- Paved the Way for Understanding the Atomic Structure
- Paved the Way for Modern Chemistry
- Impacted Our History
- Predicted the Existence of New Elements
- Provided a Systematic Way of Organizing Elements
Celebrating National Periodic Table Day is a way to acknowledge the impact of this remarkable discovery and pay tribute to the scientific community that made it possible. Similar to the National Periodic Table Day, we have recently shared many holiday posts with you. Did you read the Top 10 Reasons Why We Celebrate National Frozen Yogurt Day, Top 10 Best Carrot Cake Recipies in the World, and Top 10 Reasons Why We Celebrate World Cancer Day? So this is The Daily Top 10 Team, and we would love to invite you to write to us. We only accept the Top 10 guest posts each month. So, contact us for guest post opportunities.


Top 10 Reasons Why We Celebrate National Toast Day
Top 10 Reasons Why We Should Celebrate National Chocolate Cake Day
Top 10 Fun Things People Born in January Should Do (Updated)
Top 10 Best Scholarships in UK for International Students (Updated)
Top 10 Best & Highest-Paying In-Demand Jobs in Australia (Updated)
Top 10 Best & Most Popular Beijing Olympics Winners in 2022